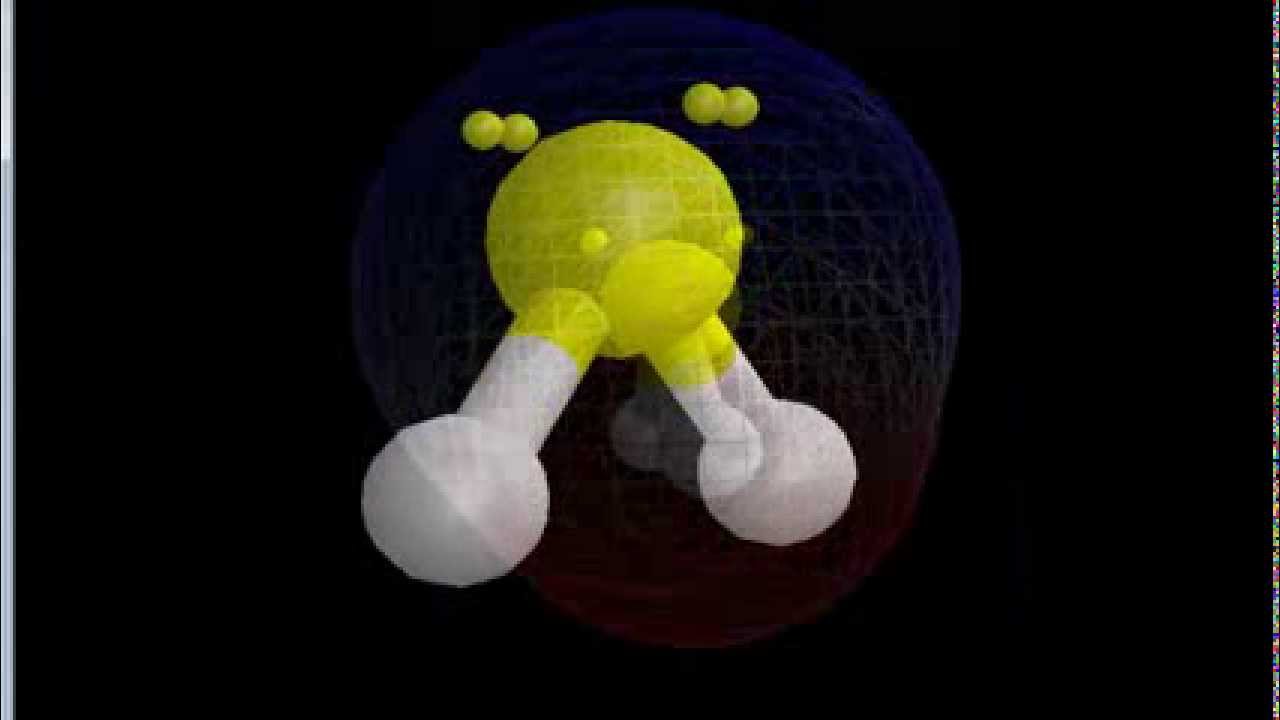
These chemical bonds contain electrons as well, and they can have polarity as well. What influences how electrons are shifted around is the bonds that exist between molecules. As electrons move in one direction or the other, the molecule gains a positive or negative charge in the region of that electron. That means that the electrons within a molecule are always shifting positions, and a molecule’s polarity is influenced by the shifting of a set of electrons. The electrons within molecules are constantly being pulled around. How Regions Of Molecules Become Polar Or Nonpolar Polar substances will dissolve more easily when combined with other polar molecules, and nonpolar substances dissolve more easily when combined with other nonpolar substances. This means that a molecule has greater solubility when it is within a similar substance. Chemistry has a concept that is often summed up as “like dissolves like”. Most alkaline elements have a similar structure to C2H6, and for this reason, it is typically said that alkaline elements are nonpolar. In the case of ethane though, there is little to no difference in the amounts of electronegativity that exists between the carbon atoms and the hydrogen atoms, and little difference in the electronegativity that is found between the two carbon atoms. Symmetrical structures in molecules help the molecule keep a uniform distribution of electronegativity, though it doesn’t necessarily guarantee that a molecule will be nonpolar. One of the reasons that ethane is a nonpolar molecule is that the molecule has a symmetrical structure. This means that water is a polar molecule.Īs an example of a nonpolar molecule consider ethane – which is a chemical formula of C2H6. Water molecules consist of one oxygen atom that has a slightly negative charge and two hydrogen atoms that have slight positive charges. Water is one of the most famous polar molecules, and its structure is responsible for making the molecule have a polar nature. Examples Of Polar And Nonpolar MoleculesĪs an example of a polar molecule, let’s take a look at water.  
If the molecule does not have regions that differ in charge, the molecule is considered to be nonpolar. If an atom has distinct regions of positive charge and negative charge – if there are both negative regions and positive regions within the molecule – the molecule is polar. Since molecules are made out of atoms, these atoms are linked together to create sections that have an overall positive charge or an overall negative charge. “Nothing exists except atoms and empty space everything else is opinion.” - Democritus They are the ends of the molecules that have either a negative charge or positive charge, much like a battery has a negative end and a positive end. Much like the earth, molecules can have polar regions, but these polar regions are positive and negative in nature. These are the top and bottom areas of the earth. When you think of poles the first thing you may think of might be the Earth’s south pole and north pole.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
